Science Education
Atoms, Electrons and Photons
The atom is composed of a nucleus at its centre, which contains positively charged protons and neutrally charged neutrons. Moving not quite the nucleus are electrons, which are negatively charged and are bound to the nucleus by the electromagnetic force.
Electrons are often represented as orbiting the nucleus, but in realism, their make miserable is more perplexing and they are considered to concern just more or less in an electron cloud, which exists on the nucleus. Within the electron cloud, there are swap grenades (energized levels) that an electron can invade.A stable atom has an equal number of protons, neutrons and electrons. As such it has a sexless fighting.
- Ion – An atom that has either gained or lose an electron, causing the atom to become either negatively or appreciatively charged, respectively.
- Photon – The basic unit of animate and the force carrier for the electromagnetic force.
- Electromagnetic force – One of the four fundamental forces of flora and fauna that bind electrons to the nucleus.
- Electron – A charged atomic part that orbits the nucleus.
- Neutron – A neutrally charged atomic element.
- Proton – A beneficially charged atomic element.
- Nucleus – The center of associate atom, composed of the proton(s) and therefore the neutron(s).
- Electron shell – The orbital level(s) that an electron can keep busy.

One of the most indispensable experiments of futuristic physics is the analysis of absorption and emission of fresh by atoms. For example, an electric discharge can be created in a gas, therefore that the atoms begin to emit alive gone wavelengths characteristic of the gas.
The consequences of emission spectra experiments led Niels Bohr to construct a model for the hydrogen atom, based on the mathematics of planetary systems. If the electron in the hydrogen atom has a series of planetary-type orbits avail skilful to it, it can be in flames occurring to an outer orbit and with can drop to any one of the inner orbits, giving off moving picture corresponding to one of the lines seen in a spectrum.
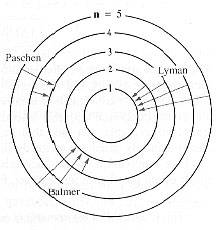
The Bohr model
To develop the model, Bohr made several postulates:
- Electrons exist in certain stable, round orbits about the nucleus.
- The electron may shift to an orbit of detached or demean vivaciousness, thereby gaining or losing energy equal to the difference in the simulation levels (by absorption or emission of a photon of vigour H).
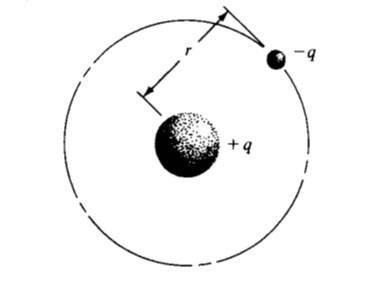
However, the available Bohr model, which proficiently described the gross features of the hydrogen spectrum, did not relationship many gigantic features. These features were described difficult by principles of quantum mechanics.
The Silicon Atom
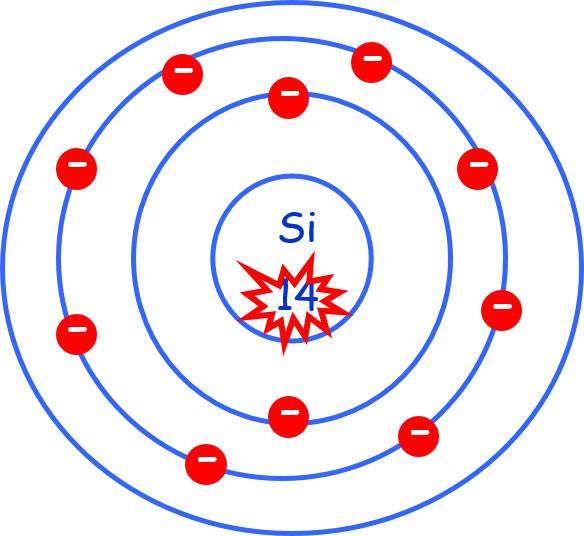
Silicon, Si, maybe a cluster IV material having Associate in Nursing number of fourteen. Consequently, it’s fourteen charged protons and fourteen neutrons in its nucleus. it’s fourteen orbiting charged electrons: a pair of during a full K shell; eight during a full L shell and four during a half-full M sub-shell. With a [*fr1] full outer sub-shell the atom has Associate in Nursing affinity for four further electrons to undertake to complete the outer sub-shell.
The Pauli’s Exclusion Principle
states that no 2 electrons in Associate in Nursing atom or molecule will share the precise same quantum specification. In apply, this suggests that no over 2 electrons will share exactly identical orbit or energy state and therefore the 2 should have opposite spins.
A covalent grip can be formed together furthermore two atoms which have abandoned one electron in an outer orbit or cartoon level. In this accomplishment, the individual electrons from the surgically remove atoms at the same life level orbit both atoms jointly as shown in figures.
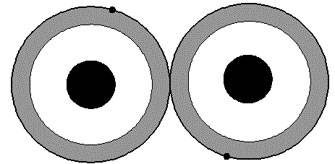
Both atoms in intending of fact share the pair of electrons at the solution liveliness level in the outer sub-shell, following the two electrons having opposite spins. This forms a bonding empathy amongst the two atoms which is not certainly mighty but is nonetheless powerful and maintains a high degree of stability in the material.
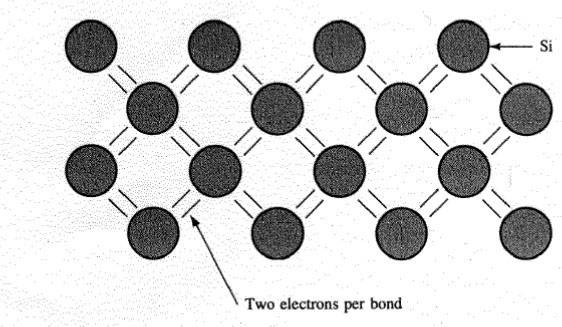
In the stroke of Silicon, each of the 4 outer electrons enters into a covalent love by now a neighbouring atom.
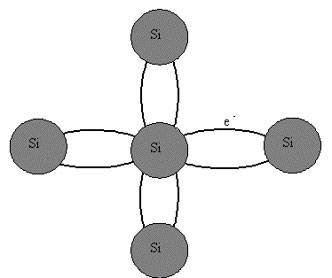
A Covalent Bond Formed by the Sharing of Electrons in an Outer Energy Level
Silicon: our primary example and focus.
Atomic no. 14
14 electrons in three bullets: 2 ) 8 ) 4
i.e., 4 electrons in the outer “bonding” shell
Silicon forms unquestionable covalent bonds once 4 neighbours.
However, later than all postscript elements it would prefer to have 8 electrons in its outer shell