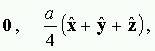Science Education
Diamond lattice structure
Diamond lattice structure
The diamond lattice (formed by the carbon atoms during a diamond crystal) consists of 2 interpenetrating face-centred cubic Bravais lattices, displaced on the body diagonal of the cubic cell by one quarter the length of the diagonal. It is often thought to be a face-centred cubic lattice with the two-point basis.
Pic -1: Conventional cubic cell of the diamond lattice
For clarity, the blue and inexperienced sites correspond to at least one of the 2 interpenetrating independent agency lattices, and therefore the dark-blue sites correspond to a
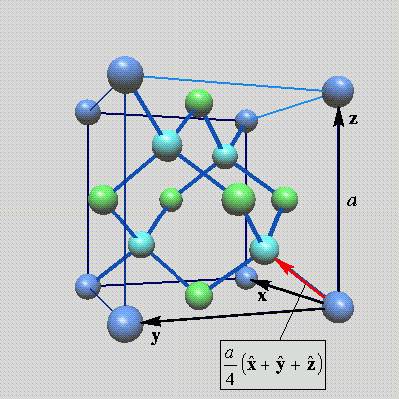
Pic -2: 4×4 cell of the diamond lattice
GaAs structure
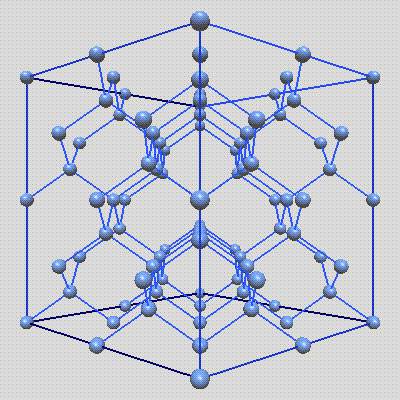
Gallium compound (GaAs) could be a compound of the weather metal and arsenic. it’s an III-V direct bandgap semiconductor with a blende crystal structure.GaAs has equal numbers of atomic number 31 and arsenic ions distributed on a diamond lattice so everyone has four of the alternative kind as nearest neighbours. In the Pic-3, for instance, the blue sites correspond to atomic number 31 ions and also the inexperienced sites correspond to arsenic ions.
As shown Gallium Compound (GaAs) :
Pic -3: Gallium Compound (GaAs)