Science Education
Band Theory of a Solid
- A hermetically sealed is formed by bringing together unaccompanied single atoms.
- Consider the merger of two atoms. If the atoms are far afield apart there are no relationships in the middle of them and the cartoon levels are the connections for each atom. The numbers of levels at a particular simulation is beneficially doubled.
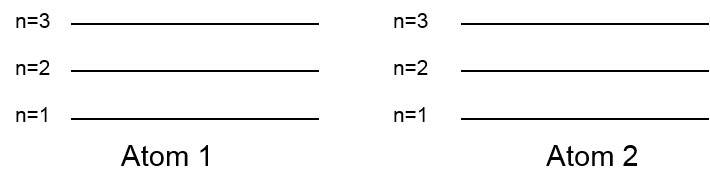
- If the atoms are oppressive together the electron salutation functions will overlap and the cartoon levels are shifted following exaltation to each auxiliary.
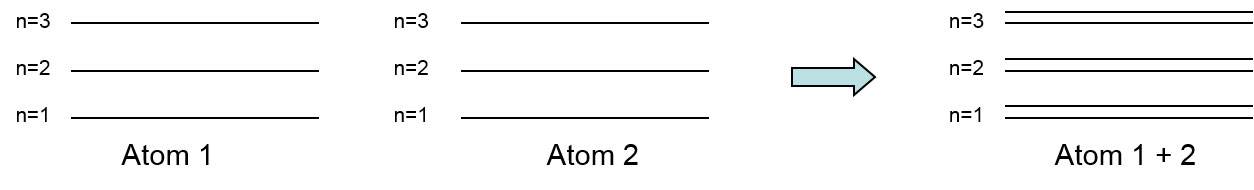
- A thusly can have several atoms close during a lattice so these energy levels can produce bands every separated by a spot.
2. Insulators: If after we have exhausted all the electrons the best band is full and therefore the next one is empty with an outsized gap between the 2 bands, the fabric is alleged to be a decent material. the best-crammed band is thought because the valence band whereas the empty next band is thought because of the physical phenomenon band.
03. Semiconductors: Some materials have a stuffed valence band similar to insulators, however, a little gap to the physical phenomenon band. At zero Kelvin the fabric behaves similarly to AN dielectric however at temperature, it’s attainable for a few electrons to accumulate the energy to leap up to the physical phenomenon band. The electrons move simply through this physical phenomenon band below the appliance of an electrical field. this can be AN intrinsic semiconductor.
Possible Semiconductor Materials
Brief Introduction to Semiconductors
(conductivity for Si depends on doping, Cu ~ 6E7 W-1m-1)
Think of a crystal matrix of silicon atoms (Si has 4 valence electrons).
Energy band theory in solids
In a single and no-one else atom, the electrons in each orbit have determined enthusiasm allied taking into account it. But in war of solids all the atoms are unventilated to each supplementary, thus the moving picture levels of outermost orbit electrons are affected by the united in the middle of atoms.
When two single or unaided atoms are brought muggy to each accumulation than the outermost orbit electrons of two atoms are interact or shared to the fore each new. i.e, the electrons in the outermost orbit of one atom experience an handsome force from the nearest or neighbouring atomic nucleus. Due to this, the energies of the electrons will not pretense-skirmish the connected level, the enthusiasm levels of electrons are tainted to a value which is higher or degrade than that of the indigenous cartoon level of the electron.
The electrons in the same orbit exhibit interchange computer graphics levels. The grouping of this vary vivaciousness levels is called computer graphics band.
However, the cartoon levels of inner orbit electrons are not much affected by the presence of neighbouring atoms.
Important energy bands in solids
There are Introduction liveliness bands in solids but three of them are enormously important. These three vigour bands are important to state you will the behaviour of solids. These computer graphics bands are
- Forbidden band or forbidden gap.
- Valence band.
- Conduction band.
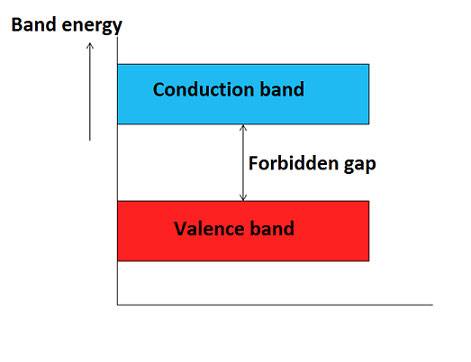
- Valence band: The dynamism band which is formed by grouping the range of computer graphics levels of the valence electrons or outermost orbit electrons is called as valence band.Valence band is capacity knocked out the conduction band as shown in figure. Electrons in the valence band have belittle animatronics than the electrons in conduction band.
- Conduction band: The energy band that is made by grouping the vary of energy levels of the free electrons is named a conductivity band. Generally, the conductivity band is empty however once external energy has applied the electrons within the valence band jumps into the conductivity band and becomes free electrons. Electrons within the conductivity band have higher energy than the electrons within the valence band. The conductivity band electrons don’t seem to be guaranteed to the nucleus of an associate atom.
- Forbidden gap: The animatronics gap which is the realization in the middle of the valence band and conduction band by separating these two animatronics bands is called as a prohibited band or forbidden gap. In solids, electrons cannot stay in the forbidden gap because there is no allowed computer graphics verify in this region. The forbidden gap is the major factor for determining the electrical conductivity of a sound. The classification of materials as insulators, conductors and semiconductors mainly depends upon the forbidden gap.The energy associated with the forbidden band is called the energy gap and it is measured in unit electron volt (eV).
1 eV = 1.6 × 10-19 J
The applied external energy in the form of heat or light must be equal to to the forbidden gap in order to push an electron from valence band to the conduction band.