Science Education
Solid State Structure
A crystalline hermetically sealed is distinguished by the fact that atoms making the crystal are decided in a periodic fashion. That is, there is some basic promise of atoms that is repeated throughout every single one sound. Thus the crystal appears exactly the related at one narrowing as it does at a series of subsidiary equivalent points, considering the basic periodicity is discovered. However, not all solids are crystals. some have no periodic structure at all (amorphous solids), and subsidiary are composed of many small regions of single-crystal material (polycrystalline solids).

The periodic contract of atoms in a crystal is called the lattice; the lattice contains a volume, called a unit cell, which is representative of every single one lattice and is regularly repeated throughout the crystal. Atoms are often gathered along as Associate in the Nursing mixture through a variety of various processes, as well as condensation, pressurization, reaction, deposit, and melting. the method sometimes determines, a minimum of at first, whether or not the gathering of atoms can go for the type of a gas, liquid or solid. The state sometimes changes as its temperature or pressure is modified. Melting is that the method most frequently accustomed type Associate in Nursing mixture of atoms. once the temperature of a soften is lowered to a precise purpose, the liquid can type either a crystalline solid or Associate in Nursing amorphous solid.
Amorphous Solids
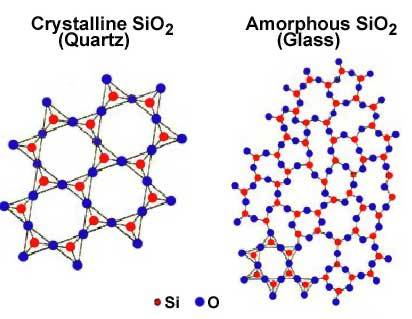
A solid substance with its atoms command apart at equilibrium spacing, however with no long-range cyclicity in atom location in its structure is associate degree amorphous solid. samples of amorphous solids square measure glass and a few styles of plastic. {they square measure|they’re} typically represented as supercooled liquids as a result of their molecules are organized in an exceedingly random manner somewhat as within the liquid state. for instance, glass is often made of silicon oxide or quartz sand, that contains a crystalline structure. once the sand is fusible and therefore the liquid is cooled quickly enough to avoid crystallization, associate degree amorphous solid known as a glass is created. Amorphous solids don’t show a pointy phase transition from solid to liquid at a precise freezing point, however rather soften step by step once they square measure heated. The physical properties of amorphous solids square measure identical all told directions on any axis in order that they square measure aforesaid to own isotropic properties, which can be mentioned in additional detail later.
The periodic contract of atoms in a crystal is called the lattice; the lattice contains a volume, called a unit cell, which is representative of every single one lattice and is regularly repeated throughout the crystal. Atoms are often gathered along as Associate in the Nursing mixture through a variety of various processes, as well as condensation, pressurization, reaction, deposit, and melting. the method sometimes determines, a minimum of at first, whether or not the gathering of atoms can go for the type of a gas, liquid or solid. The state sometimes changes as its temperature or pressure is modified. Melting is that the method most frequently accustomed type Associate in Nursing mixture of atoms. once the temperature of a soften is lowered to a precise purpose, the liquid can type either a crystalline solid or Associate in Nursing amorphous solid.
Crystalline Solids
More than ninetieth of present and by artificial means ready solids square measure crystalline. A crystal could be a regular, continuance arrangement of atoms or molecules. the bulk of solids, as well as all metals, adopt a crystalline arrangement as a result of the number of stabilization achieved by anchoring interactions between neighbouring particles is at its greatest once the particles adopt regular (rather than random) arrangements. within the crystalline arrangement, the particles pack expeditiously along to attenuate the overall building block energy.
The regular continuance pattern that the atoms prepare its named the crystalline lattice. The scanning tunnelling magnifier (STM) makes it attainable to image the lepton cloud associated individual atoms at the surface of a cloth. Below is AN immediate memory image of a Pt surface showing the regular alignment of atoms.
Fig: BM Research, Almaden Research Center.
Crystal structures may be conveniently specified by describing the arrangement within the solid of a small representative group of atoms or molecules, called the ‘unit cell.’ By multiplying identical unit cells in three directions, the location of all the particles in the crystal is determined. In nature, 14 different types of crystal structures or lattices are found. The simplest crystalline unit cell to picture is the cubic, where the atoms are lined up in a square, 3D grid. The unit cell is simply a box with an atom at each corner.
Unit Cells for Types of Cubic lattice structure.
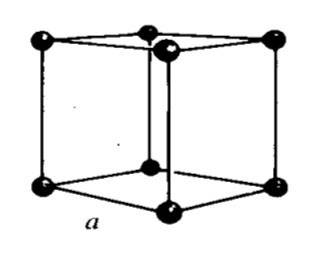
Fig : Simple cubic (sc)
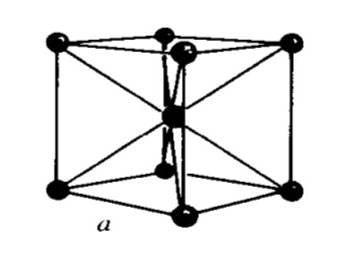
Fig : Body-centered cubic (BCC)
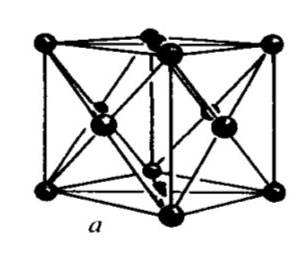
Fig : Face-centered cubic (FCC)
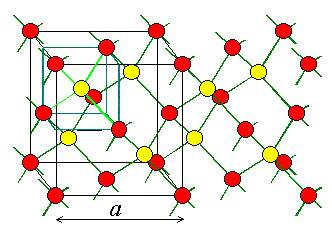
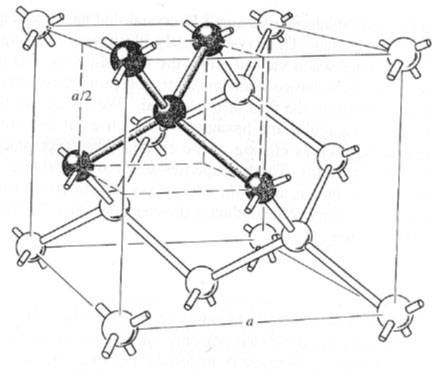
Diamond lattice unit cell, showing the four nearest neighbour structure
The basic lattice structure for several vital semiconductors is that the diamond lattice, that is characteristic of Si and Ge. In several compound semiconductors, atoms are organized in a very basic diamond structure, however, are alternating in a bank account to rotate sites. this is often known as a zincblende lattice and is typical of the III-V compounds. The diamond lattice is thought of as associate degree independent agency structure taking into thought associate degree adding happening atom placed at a/4+b/4+c/4 from every of the independent agency atoms.
Each atom within the diamond lattice encompasses a chemical bond with four adjacent atoms, that along kinda polyhedron. This lattice may be shaped from 2 fcc-cubic lattices, that are displayed on the body diagonal of the larger cube in Figure by one-quarter of that body diagonal. The diamond lattice thus could be an fcc-cubic lattice with a basis containing 2 identical atoms. Extended valency (covalent network solids) solids incorporate atoms that ar control along in giant networks containing extended valency bonds. Quartz is Associate in Nursing example of a 3-dimensional network of valence bonds. (See pages 873 – 879 in BLB.) Quartz consists of SiO2 teams that covalently bond to alternative SiO2 teams all told 3 directions. Notice that each one the semiconducting material atom ar tetrahedral guaranteed to element atoms. In diamond (see Figure eleven.40(a) on page 403), that is one style of elemental carbon, all the carbon atoms are tetrahedrally guaranteed in a very 3-dimensional network of valence bonds. black lead (see Figure eleven.40(b) on page 403) consists of sheets of a hexangular guaranteed, sp2 hybridized, carbon atoms. the very fact that these sheets are bedded on high of every alternative permit for slippage of layers relative to every alternative creating black lead a superb material.
Fig : Diamond lattice of silicon and germanium.
Each atom within the diamond lattice encompasses a chemical bond with four adjacent atoms, that along kinda polyhedron. This lattice may be shaped from 2 fcc-cubic lattices, that are displayed on the body diagonal of the larger cube in Figure by one-quarter of that body diagonal. The diamond lattice thus could be an fcc-cubic lattice with a basis containing 2 identical atoms. Extended valency (covalent network solids) solids incorporate atoms that ar control along in giant networks containing extended valency bonds. Quartz is Associate in Nursing example of a 3-dimensional network of valence bonds. (See pages 873 – 879 in BLB.) Quartz consists of SiO2 teams that covalently bond to alternative SiO2 teams all told 3 directions. Notice that each one the semiconducting material atom ar tetrahedral guaranteed to element atoms. In diamond (see Figure eleven.40(a) on page 403), that is one style of elemental carbon, all the carbon atoms are tetrahedrally guaranteed in a very 3-dimensional network of valence bonds. black lead (see Figure eleven.40(b) on page 403) consists of sheets of a hexangular guaranteed, sp2 hybridized, carbon atoms. the very fact that these sheets are bedded on high of every alternative permit for slippage of layers relative to every alternative creating black lead a superb material.